In
The Hatch-Waxman Patent Term Extension Statute
The patent term extension provisions of the Hatch-Waxman Act are set forth in 35
(a)The term of a patent which claims a product, a method of using a product, or a method of manufacturing a product shall be extended in accordance with this section from the original expiration date of the patent, which shall include any patent term adjustment granted under section 154(b), if— ...
(4) the product has been subject to a regulatory review period before its commercial marketing or use ....
The extended rights are set forth in § 156(b), which provides in relevant part that, for a patent which claims a method of using a product, the extended rights are "limited to any use claimed by the patent and approved for the product."
At issue in this case was the meaning of the term "product," which § 156(f) defines as a "drug product," which it in turn defines as the "active ingredient" of a new drug "including any salt or ester of the active ingredient, as a single entity or in combination with another active ingredient."
The Patent And Accused Product At Issue
The patent at issue was Biogen's
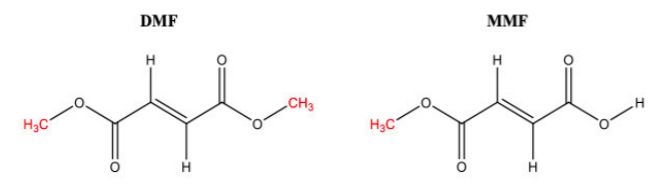
In chemical terms. MMF can be described as a de-esterified form of the double ester DMF. The Federal Circuit opinion depicts their structures as follows:
The court treated claim 1 of the '001 patent as representative:
- A method of treating multiple sclerosis comprising administering, to a patient in need of treatment for multiple sclerosis, an amount of a pharmaceutical preparation effective for treating multiple sclerosis, the pharmaceutical preparation comprising at least one excipient or at least one carrier or at least one combination thereof; and dimethyl fumarate, methyl hydrogen fumarate, or a combination thereof.
Claim 1 literally encompasses both DMF and MMF.
The Federal Circuit Decision
The Federal Circuit decision was authored by
The court considered the parties case law-based arguments, but determined that the issue before it "is governed by the statute." In particular, the court focused on the "active ingredient" definition of "product" in § 156(f), stating:
"Active ingredient" is a term of art, defined by the FDA as "any component that is intended to furnish pharmacological activity or other direct effect," 21 C.F.R. § 210.3(b)(7), and it "must be present in the drug product when administered."
As noted above, § 156(f) defines "drug product" as "including any salt or ester of the active ingredient." Applying these definitions to the facts before it, the court stated:
MMF is not the approved product, nor is it specified as the active ingredient on the Tecfidera® label. .... MMF is the de-esterified form of DMF, not an ester of DMF.
Thus, the court reasoned, Biogen's extended patent rights based on FDA approval of DMF do not reach MMF, because:
[T]he term "product," defined in § 156(f) as the "active ingredient . . . including any salt or ester of the active ingredient," has a plain and ordinary meaning that is not coextensive with "active moiety." It encompasses the active ingredient that exists in the product as administered and as approved—as specified by the FDA and designated on the product's label—or changes to that active ingredient which serve only to make it a salt or an ester. It does not encompass a metabolite of the active ingredient or its de-esterified form. This case is unlike Glaxo or
The Federal Circuit also held that Biogen could not use the doctrine of equivalents to establish infringement:
To infringe a patent claim extended under § 156, an accused product or process must meet, either literally or through equivalence, each individual element of the claim. See Johnson & Johnston Assocs. Inc. v.
What Did
Biogen argued that its position was supported by the "active moiety" analysis in
The FDA ruled that "the term 'active ingredient' as used in the phrase 'active ingredient including any salt or ester of the active ingredient' means active moiety." Abbreviated New Drug Application Regulations: Patent and Exclusivity Provisions, 59 Fed. Reg. 50,338, 50,358 (F.D.A.
(The FDA rule cited in
In
As Biogen pointed out in its petition for rehearing en banc, however, the approved product in
The content of this article is intended to provide a general guide to the subject matter. Specialist advice should be sought about your specific circumstances.
Ms
IL 60654-5313
Tel: 312832 4500
Fax: 312832 4700
E-mail: info@foley.com
URL: www.foley.com
© Mondaq Ltd, 2020 - Tel. +44 (0)20 8544 8300 - http://www.mondaq.com, source









