Preliminary OvaWatch® volume during Q2 2024 grew 24% over the prior quarter to 1,307 units
Preliminary OvaSuiteSM volume during Q2 2024 grew 11% over the prior quarter to 6,471 units
Preliminary Second Quarter 2024 Product Volume and Highlights
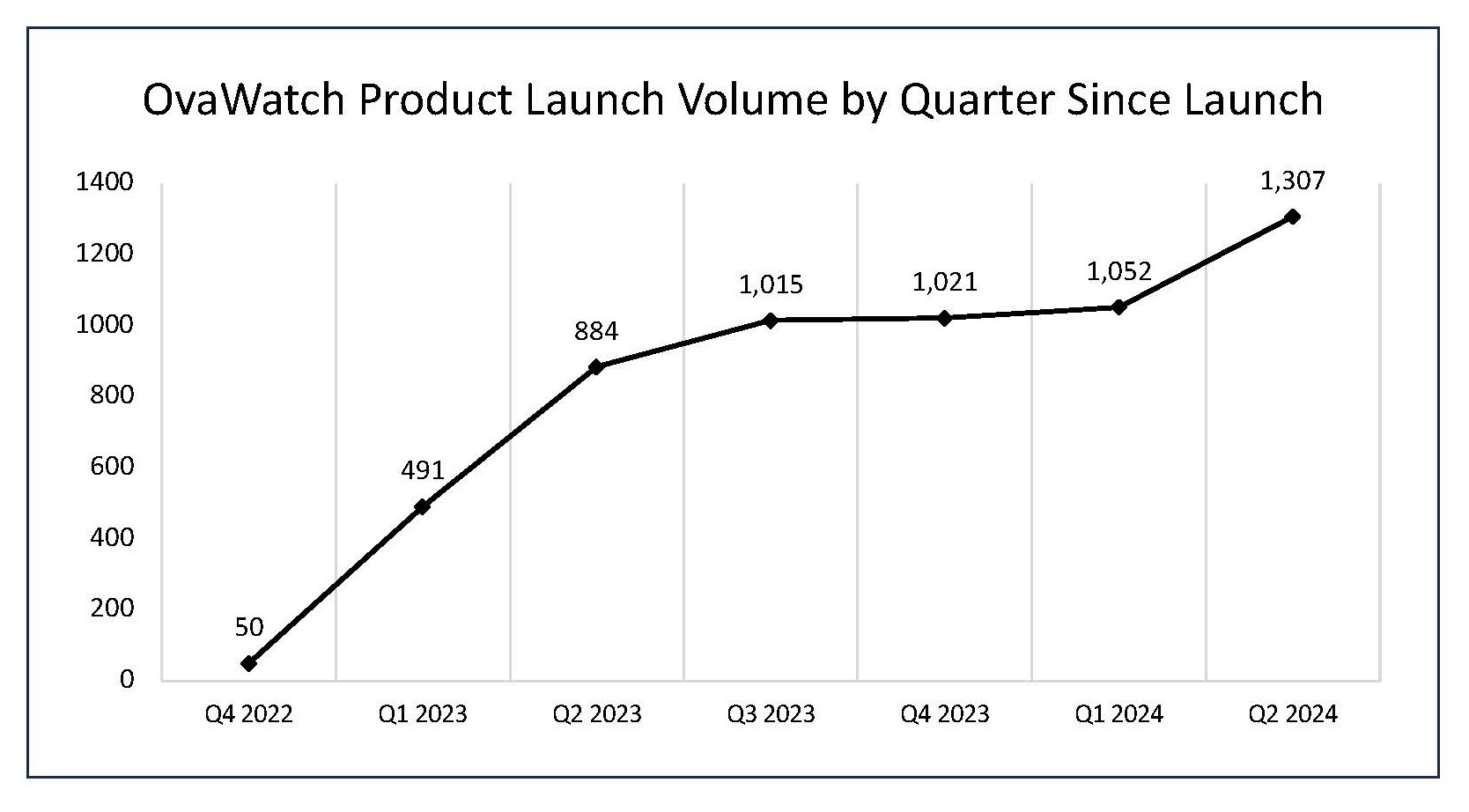
- The number of OvaWatch tests performed during the second quarter ended
June 30, 2024 , was 1,307, an increase of 48%, compared to the 884 tests in the same period last year, and an increase of 24% to the 1,052 tests sequentially.
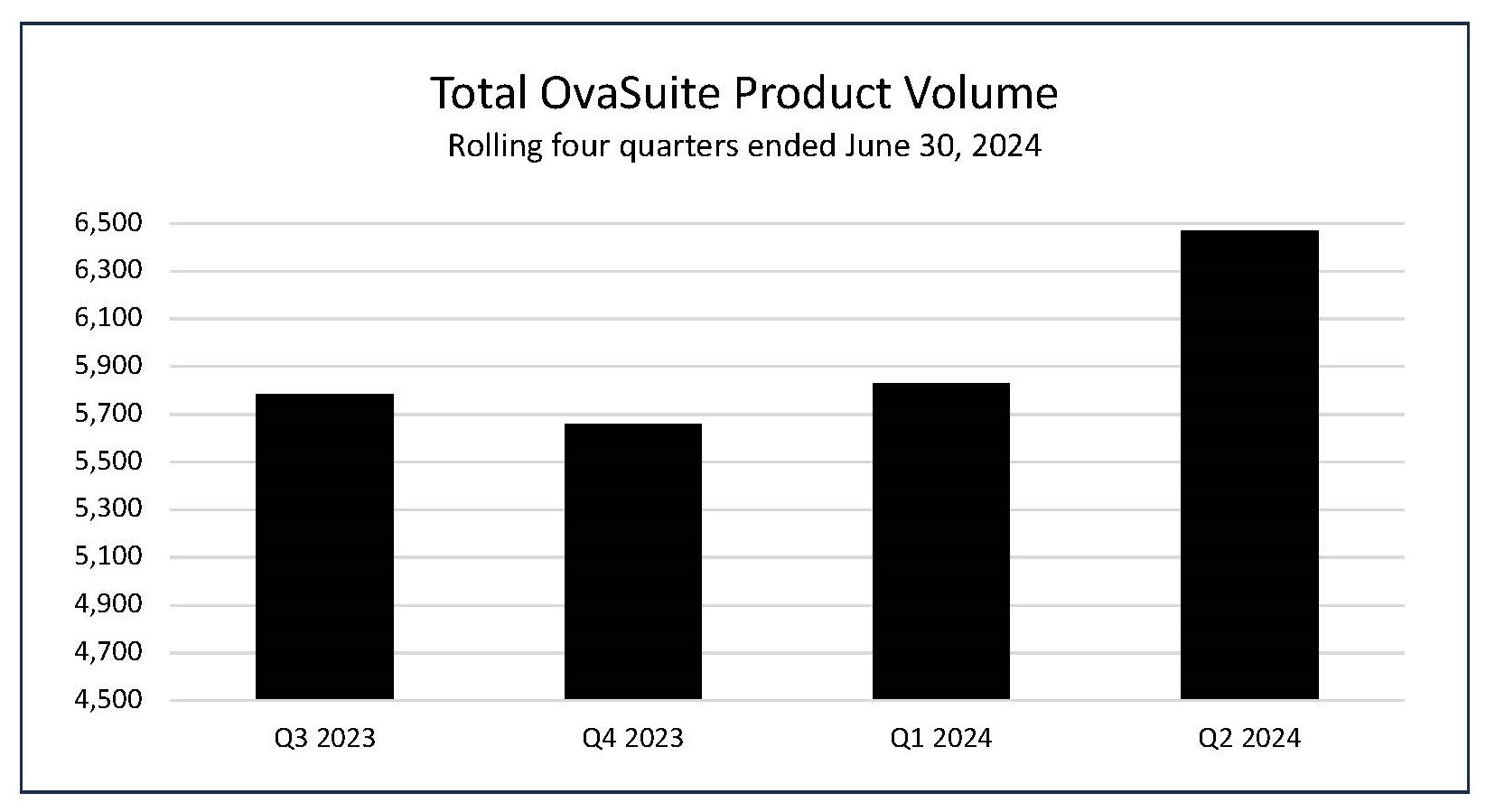
- The number of OvaSuite tests performed during the second quarter ended
June 30, 2024 , was 6,471, an increase of 3% compared to the 6,289 tests in the same period last year, and an increase of 11% to the 5,829 tests in the first quarter.
- The Company was notified that it has been selected as a finalist for an ARPA-H grant to support further development of its protein + microRNA blood test for endometriosis. Announcement of the final grant award recipients is anticipated in the second half of 2024.
- The Company has completed a comprehensive analysis of its biobank and has identified up to 70,000 serum, plasma, and whole blood samples that are available for secondary research. The Company is exploring opportunities to create non-dilutive sources of cash through related collaborations.
“Our OvaSuite product volume, and in particular our OvaWatch volume, has grown each month since the beginning of the year, demonstrating that the commercial changes we made last year are having a positive impact,” said
Dr.
“With respect to our in-development pipeline, we continue to progress in the design and development of our molecular tests,”
In addition to these second-quarter highlights, the Company recently completed a
About Aspira Women’s
Aspira Women’s
OvaWatch® and Ova1Plus® are offered to clinicians as OvaSuiteSM. Together, they provide the only comprehensive portfolio of blood tests to aid in the detection of ovarian cancer for the 1.2+ million American women diagnosed with an adnexal mass each year. OvaWatch provides a negative predictive value of 99% and is used to assess ovarian cancer risk for women where initial clinical assessment indicates the mass is indeterminate or benign, and thus surgery may be premature or unnecessary. Ova1Plus is a reflex process of two FDA-cleared tests, Ova1® and Overa®, to assess the risk of ovarian malignancy in women planned for surgery.
Our in-development test pipeline is designed to expand our ovarian cancer portfolio and addresses the tremendous need for noninvasive diagnostics for endometriosis, a debilitating disease that impacts millions of women worldwide. In ovarian cancer, our OvaMDxSM risk assessment is designed to combine microRNA and protein biomarkers with patient data to further enhance the sensitivity and specificity of our current tests. In endometriosis, EndoCheckSM is the first-ever noninvasive test designed to identify endometriomas, one of the most commonly occurring forms of endometriosis. The EndoMDxSM test is designed to combine microRNA and protein biomarkers with patient data to identify all endometriosis.
Forward-Looking Statements
This press release contains forward-looking statements, as defined in the Private Securities Litigation Reform Act of 1995. Forward-looking statements involve a number of risks and uncertainties. Such forward-looking statements include statements regarding, among other things, the timing and completion of any products in the pipeline development and other statements that are predictive in nature. Actual results could differ materially from those discussed due to known and unknown risks, uncertainties, and other factors. These forward-looking statements generally can be identified by the use of words such as “designed to,” “expect,” “plan,” “anticipate,” “could,” “may,” “intend,” “will,” “continue,” “future,” other words of similar meaning and the use of future dates. Forward-looking statements in this press release and other factors that may cause such differences include the satisfaction of customary closing conditions related to the offering and the expected timing of the closing of the offering. These and additional risks and uncertainties are described more fully in the company’s filings with the
Investor Relations Contact:
Nicole Sandford
Chief Executive Officer
Investors@aspirawh.com
Photos accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/839c39d0-429f-436e-aaff-beed552ac624
https://www.globenewswire.com/NewsRoom/AttachmentNg/bcdf6e34-66dc-4d38-b217-3e2bc390ca7d

![]()
Figure 1
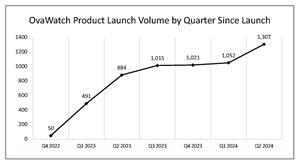
OvaWatch Product Launch Volume by Quarter Since Launch
Figure 2
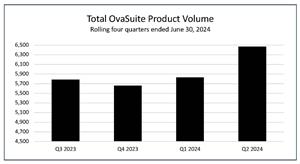
Total OvaSuite Product Volume
2024 GlobeNewswire, Inc., source







