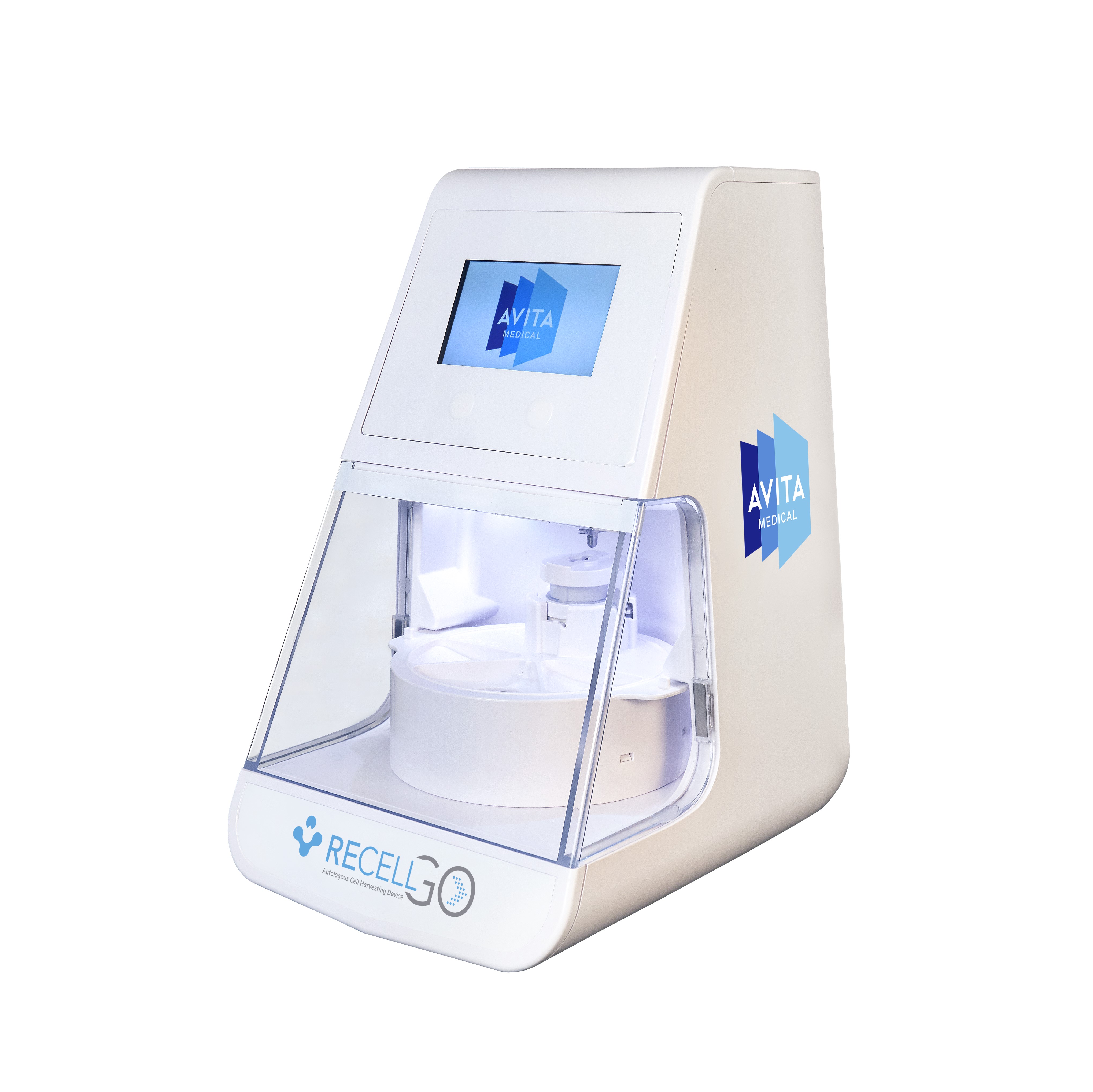
When choosing RECELL, clinicians and patients can realize several significant advantages over traditional skin grafting:
- Improved healing is achieved using significantly less donor skin1
- Pain is reduced, closure is faster, and the aesthetic appearance at the RECELL-harvested donor site is improved2
- Fewer procedures are required for definitive closure3
- There's a reduction in the length of stay for burns covering less than 50% Total Body Surface Area (TBSA)2,3,4
RECELL GO introduces enhanced features that streamline the preparation of Spray-On Skin™ Cells. This next-generation device significantly reduces the training burden on medical staff, improves workflow efficiency in the operating room, and controls the RECELL Enzyme™ incubation time to ensure optimal cell yield and viability. These advancements simplify the user interface, enabling medical teams to provide quality care readily and consistently to their patients.
"FDA approval of RECELL GO marks a paradigm shift in the treatment of partial-thickness and full-thickness wounds,” said
In
The supplement follows the original PMA of RECELL Autologous Cell Harvesting Device and subsequent PMA supplements.
About
In international markets, the RECELL System is approved to promote skin healing in a wide range of applications including burns, full-thickness skin defects, and vitiligo. The RECELL System is TGA-registered in
To learn more, visit www.avitamedical.com.
Forward-Looking Statements
Statements in this announcement may contain forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Such forward-looking statements are subject to significant risks and uncertainties that could cause actual results to differ materially from those expressed or implied by such statements. Forward-looking statements generally may be identified by the use of words such as “anticipate,” “expect,” “intend,” “could,” “may,” “will,” “believe,” “estimate,” “look forward,” “forecast,” “goal,” “target,” “project,” “continue,” “outlook,” “guidance,” “future,” and similar words or expressions, and the use of future dates. Forward-looking statements in this announcement include but are not limited to statements concerning our product development activities, regulatory approval of our products, the potential for future growth of our business, and our ability to achieve financial goals. These statements are made as of the date of this announcement, and the Company undertakes no obligation to publicly update or revise any of these statements, except as required by law. For additional information and other important factors that may cause actual results to differ materially from forward-looking statements, please see the “Risk Factors” section of the Company’s latest Annual Report on Form 10-K and other publicly available filings for a discussion of these and other risks and uncertainties.
Authorized for release by the Chief Financial Officer of
____________________
1 Instructions for Use. RECELL® Autologous Cell Harvesting Device.
2 Holmes JH, Molnar JA, Carter JE, et al. A comparative study of the RECELL® device and autologous split-thickness meshed skin graft in the treatment of acute burn injuries. J Burn Care Res. 2018;39(5):694-702.
3 Kowal S, Kruger E, Bilir P, et al. Cost effectiveness of the use of autologous cell harvesting device compared to standard of care for treatment of severe burns in
4 Holmes JH, Molnar JA, Carter JE, et al. A comparative study of the RECELL® device and autologous split-thickness meshed skin graft in the treatment of acute burn injuries. J Burn Care Res. 2018;39(5):694-702.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/5e843520-955d-4f45-a7f8-53b0a6f3a3f6

Investor & Media Contact:Jessica Ekeberg Phone +1-661-904-9269 investor@avitamedical.com media@avitamedical.com

Introducing RECELL GO™: Revolutionizing Wound Care
The RECELL GO™ System by AVITA Medical : A next-generation autologous cell harvesting device that harnesses the regenerative properties of a patient’s own skin to create Spray-On Skin™ Cells, delivering transformative point-of-care in the treatment of thermal burn wounds and full-thickness skin defects.
2024 GlobeNewswire, Inc., source







